
|

|

|

|
|
This article may be reprinted free of charge provided 1) that there is clear attribution to the Orthomolecular Medicine News Service, and 2) that both the OMNS free subscription link http://orthomolecular.org/subscribe.html and also the OMNS archive link http://orthomolecular.org/resources/omns/index.shtml are included.
The Nutrient Demand Principle of DiseaseWhy illness and aging increase the physiological requirement for optimal nutritionby Richard Z. Cheng, MD, PhD
Modern medicine often assumes that when organs begin to fail, the safest strategy is to restrict nutritional intake, particularly vitamins and minerals that might accumulate in the body. Patients with chronic kidney disease, heart failure, or advanced age are frequently advised to limit certain nutrients. While this cautious approach may appear reasonable from a pharmacological perspective, it may overlook a fundamental physiological reality: Disease increases metabolic stress, and metabolic stress increases nutrient demand. This relationship can be summarized as a physiological principle: IOM Nutrient Demand PrincipleThe greater the biological stress or disease burden, the greater the physiological requirement for optimal concentrations of essential nutrients. This relationship is illustrated conceptually in Figure 1. 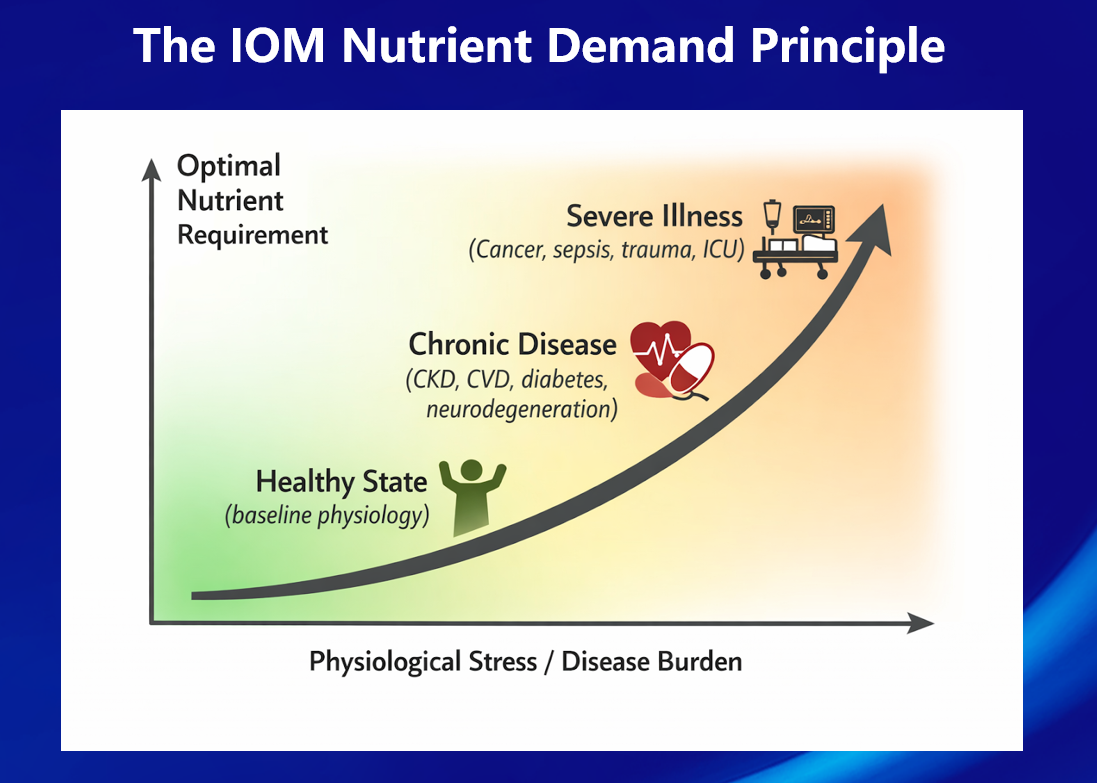
Figure 1. The IOM Nutrient Demand Principle. Optimal physiological requirements for essential nutrients increase as biological stress and disease burden rise-from baseline health to chronic disease and severe illness. This principle is consistent with fundamental concepts in physiology: biological stress increases metabolic turnover, and increased metabolic turnover requires greater biochemical resources. Biological Stress Increases Metabolic RequirementsMany disease processes activate a common set of metabolic responses, including:
These processes increase biochemical turnover and metabolic demand. A large body of research indicates that oxidative stress and mitochondrial dysfunction are important mechanisms underlying many chronic diseases, including cardiovascular disease, chronic kidney disease, diabetes, and neurodegeneration [1-3]. These processes require nutrients that function as metabolic cofactors. For example:
When metabolic stress increases, utilization of these nutrients rises accordingly. Chronic Disease and Micronutrient DepletionNumerous studies demonstrate that patients with chronic disease frequently exhibit micronutrient deficiencies. Patients with chronic kidney disease, for example, often show reduced levels of vitamin C, vitamin D, zinc, and selenium [8-10]. Dialysis further contributes to depletion of water-soluble vitamins through extracorporeal loss [11]. Similarly, magnesium deficiency has been associated with cardiovascular disease and metabolic syndrome [12], while vitamin D deficiency has been linked to immune dysregulation and chronic inflammatory disorders [13]. Hospitalized and critically ill patients frequently develop rapid micronutrient depletion due to increased metabolic demand combined with reduced intake and impaired absorption [14]. Thus, paradoxically, the patients who are most ill often have the lowest physiological reserves of essential nutrients. Aging Amplifies the ProblemAging further increases the importance of optimal nutrition. Older adults often experience:
These biological changes increase vulnerability to disease while simultaneously increasing metabolic requirements for micronutrients. Studies of aging populations have consistently demonstrated widespread micronutrient insufficiencies, particularly involving vitamin D, magnesium, and several B vitamins [15]. The Paradox of Modern Clinical NutritionDespite growing evidence of micronutrient depletion in chronic disease, clinical practice often emphasizes nutrient restriction rather than nutrient optimization. For example, patients with renal insufficiency are frequently advised to restrict vitamin C or other nutrients due to concerns about metabolic accumulation. While safety considerations are important, excessive restriction may contribute to chronic under-nutrition, particularly in patients who already have depleted nutrient reserves. This creates a paradox: Those with the highest metabolic demand may receive the least nutritional support. Why Restriction Models Persist in MedicineThe persistence of nutrient restriction models is partly historical. Modern medical training developed within a drug-centered paradigm, in which administered substances are treated primarily as pharmacologic agents that may accumulate or cause toxicity. While this framework is appropriate for many drugs, it is less applicable to substances that are essential components of normal physiology. Vitamins and minerals are not foreign chemicals but intrinsic biochemical participants in metabolism. Their optimal concentrations may vary depending on metabolic demand, physiological stress, and individual biochemical variation. Determining Optimal Nutrient IntakeThe optimal intake of vitamins and minerals is not identical for all individuals. Requirements vary according to age, body composition, metabolic stress, disease burden, and individual biochemical variation. In clinical practice, several laboratory measurements may help guide nutritional optimization, including:
These measurements can provide useful information about nutritional status and metabolic stress. However, it is important to recognize that many conventional laboratory tests may underestimate intracellular deficiencies. For example, serum magnesium levels often remain within the "normal" range even when total body magnesium stores are depleted. Therefore, clinical evaluation of nutritional status often requires integration of laboratory data, clinical symptoms, dietary history, and therapeutic response. From Drug-Centered Medicine to Metabolic MedicineModern biomedical research increasingly recognizes the importance of metabolism in chronic disease. Conditions such as cardiovascular disease, diabetes, neurodegeneration, and cancer share several common biological mechanisms, including:
Optimal nutrient availability plays a central role in regulating these systems. This understanding suggests the need for a broader medical framework that recognizes metabolic support and nutritional optimization as fundamental components of disease management. Nutrient Demand Is DynamicAnother important implication of the Nutrient Demand Principle is that optimal nutrient intake is dynamic rather than fixed. When a deficiency exists, the body may initially utilize nutrients rapidly to restore depleted tissues and metabolic systems. During this phase, higher intake levels may be required. Magnesium provides a useful illustration. Individuals with magnesium deficiency may tolerate relatively high oral intake without adverse effects as tissues and bones gradually replenish their magnesium stores. Once physiological stores are restored, the same intake level may exceed current requirements and lead to increased excretion or mild laxative effects. Vitamin C demonstrates a similar phenomenon. During periods of infection, surgery, or severe physiological stress, the body may require substantially higher intakes to support immune function and antioxidant defense. As recovery progresses and metabolic stress decreases, the optimal intake level typically declines. Thus, optimal nutrient intake should be understood as a dynamic physiological requirement that varies with health status, metabolic stress, and recovery. Increased Nutrient Demand During Acute InfectionAcute infections provide a clear illustration of the Nutrient Demand Principle. During infection, metabolic stress increases substantially due to immune activation, oxidative stress, inflammatory signaling, and tissue repair. These processes can significantly increase the physiological requirement for several micronutrients. For example, orthomolecular clinical practice has long recognized that vitamin C requirements may increase markedly during acute infections. Oral vitamin C intake may range from several grams per day in healthy individuals to divided doses totaling 10-20 grams or more per day, adjusted to gastrointestinal tolerance, during periods of acute illness. As the infection resolves and metabolic stress declines, the optimal intake typically decreases. Other nutrients may show similar demand-dependent variation. During periods of acute illness clinicians may temporarily employ higher intake levels such as vitamin D (e.g., short-term loading doses followed by several thousand IU daily), magnesium (approximately 1-2 grams per day depending on tolerance), and zinc (often 50-100 mg per day for short durations) to support immune function and metabolic resilience. Increased nutrient demand is not limited to acute infections. Aging, chronic diseases, toxic exposures, and sustained metabolic stress may also increase the physiological requirement for key micronutrients involved in mitochondrial metabolism, antioxidant defense, immune regulation, and tissue repair. Dose requirements may also vary according to age, body size, baseline nutritional status, absorptive capacity, and overall disease burden. These examples illustrate an important physiological principle: optimal nutrient intake is not static. Requirements may increase substantially during periods of physiological stress-such as infection, trauma, chronic disease, or aging-and may decline again as metabolic balance and tissue stores are restored. Thus, the optimal intake of micronutrients should be understood as context-dependent and dynamic, rather than fixed at a universal daily amount. For a more detailed clinical discussion of orthomolecular nutritional strategies during acute infections, see: "Treating Infections: An Integrative Orthomolecular Medicine Protocol" ConclusionThe IOM Nutrient Demand Principle highlights an important physiological reality: Illness is metabolically expensive. Healing requires energy, antioxidant defense, immune competence, and tissue regeneration-processes that depend on adequate nutritional resources. Recognizing the IOM Nutrient Demand Principle may help guide medicine toward a more physiological approach-one that supports metabolic resilience and the body's intrinsic capacity for healing. References1. Finkel T, Holbrook NJ. Oxidants, oxidative stress and the biology of ageing. Nature. 2000;408:239-247. 2. Wallace DC. A mitochondrial paradigm of metabolic and degenerative diseases, aging, and cancer. Annual Review of Genetics. 2005;39:359-407. 3. Stenvinkel P. Inflammation in end-stage renal disease. Nephrology Dialysis Transplantation. 2010;25:1623-1628. 4. Carr AC, Maggini S. Vitamin C and immune function. Nutrients. 2017;9:1211. 5. de Baaij JHF, Hoenderop JGJ, Bindels RJM. Magnesium in man: implications for health and disease. Physiological Reviews. 2015;95:1-46. 6. Kennedy DO. B vitamins and the brain: mechanisms, dose and efficacy-A review. Nutrients. 2016;8:68. 7. Rayman MP. Selenium and human health. Lancet. 2012;379:1256-1268. 8. Himmelfarb J, Stenvinkel P. Oxidative stress in chronic kidney disease. Journal of the American Society of Nephrology. 2005;16:S20-S24. 9. Morena M, Cristol JP, Bosc JY, Tetta C, Forret G, Leger CL, Delcourt C, Papoz L, Descomps B, Canaud B. Convective and diffusive losses of vitamin C during haemodiafiltration sessions. Nephrology Dialysis Transplantation. 2002;17:422-427. 10. Tonelli M, Wiebe N, Hemmelgarn B, Klarenbach S, Field C, Manns B, Thadhani R, Gill J. Trace elements in hemodialysis patients: a systematic review and meta-analysis. BMC Medicine. 2009;7:25. 11. Clase CM, Ki V, Holden RM. Water-soluble vitamins in people with low glomerular filtration rate or on dialysis. Seminars in Dialysis. 2013;26:546-567. 12. DiNicolantonio JJ, O'Keefe JH, Wilson W. Subclinical magnesium deficiency: a principal driver of cardiovascular disease. Open Heart. 2018;5:e000668. 13. Grant WB, Wimalawansa SJ, Pludowski P, Cheng RZ. Vitamin D: Evidence-based health benefits and recommendations for population guidelines. Nutrients. 2025;17:277. 14. Berger MM, Shenkin A. Update on clinical micronutrient supplementation studies in the critically ill. Current Opinion in Clinical Nutrition and Metabolic Care. 2006;9:711-716. 15. ter Borg S, Verlaan S, Hemsworth J, Mijnarends DM, Schols JMGA, Luiking YC, de Groot LCPGM. Micronutrient intakes and potential inadequacies of community-dwelling older adults: a systematic review. British Journal of Nutrition. 2015;113:1195-1206. Orthomolecular MedicineOrthomolecular medicine uses safe, effective nutritional therapy to fight illness. For more information: http://www.orthomolecular.org Find a DoctorTo locate an orthomolecular physician near you: http://orthomolecular.org/resources/omns/v06n09.shtml The peer-reviewed Orthomolecular Medicine News Service is a non-profit and non-commercial informational resource. Editorial Review Board:
Jennifer L. Aliano, M.S., L.Ac., C.C.N. (USA)
Comments and media contact: editor@orthomolecular.org OMNS welcomes but is unable to respond to individual reader emails. Reader comments become the property of OMNS and may or may not be used for publication. To Subscribe at no charge: https://www.orthomolecular.org/subscribe.html To Unsubscribe from this list: https://www.orthomolecular.org/unsubscribe.html |

This website is managed by Riordan Clinic
A Non-profit 501(c)(3) Medical, Research and Educational Organization
3100 North Hillside Avenue, Wichita, KS 67219 USA
Phone: 316-682-3100; Fax: 316-682-5054
© (Riordan Clinic) 2004 - 2024c
Information on Orthomolecular.org is provided for educational purposes only. It is not intended as medical advice.
Consult your orthomolecular health care professional for individual guidance on specific health problems.